Ticker: MSCL (TSX) and MSCLF (OTC)
Market cap: 64.29MCAD
Canadian small/microcap Biotech company on the verge of human trials. Doesn’t that sound good?
Founded in 2018. Listed on the stock exchange (TSXV) in 2021 via a reverse takeover. I personally invested in this around that time and have increased my position over the years. I am not a medical expert!
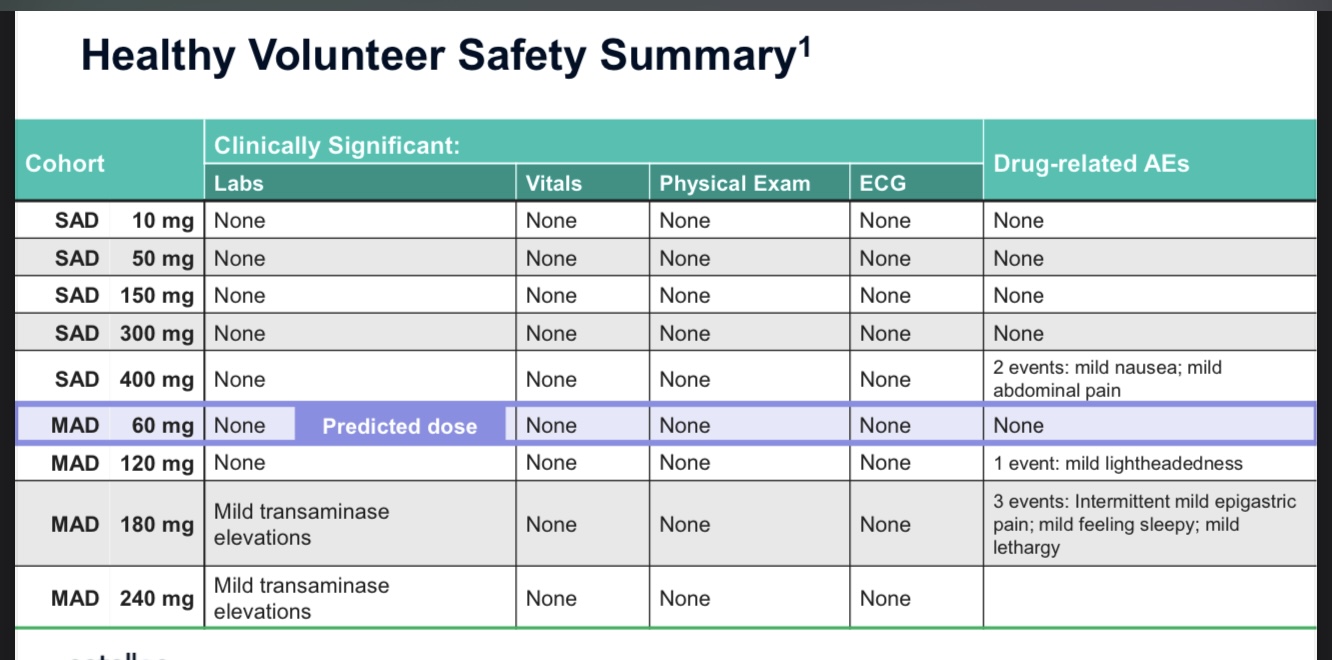
The company has communicated that it will start Phase 1 human trials in the summer. It has cash for two years. Approx. 40M CAD.
Listing on Canada’s main stock exchange (TSX) happened recently. On the US side, listing on Nasdaq is the goal for this year.
Latest presentation https://ir.satellos.com/files/doc_downloads/2024/02/14/Satellos-Deck_Feb-2024.pdf
In brief:
Developing an oral drug for Duchenne muscular dystrophy (Lead program). The prevalence of this genetic disease is globally consistent, with an incidence of approximately 30/100,000 male births. The disease currently leads to the death of boys at 20-30 years of age. (Duchennen lihasdystrofia – Wikipedia).
Lead Program: SAT-3247
Pipeline/Technologies
- The treatment of Duchenne muscular dystrophy via muscle regeneration.
- FSHD - Muscle function in FSHD mouse model improves with SAT-3247
- The treatment of other muscular dystrophies (LAMA2, Collagen-VI, Limb Girdle MD (LGMD##) and Dystroglycanopathies)
- MyoReGenX - discovery platform, two discoveries are undisclosed at this point, discovered the aged muscle
treatment opportunity - OralTrans - oral delivery of non-water soluble compounds, dozens of use cases
- JDA with NW PharmaTech (Announced March 11, 2022 - news on the joint venture corporation to follow)
- OralTrans delivery
- Drug development targeted to improve muscle quality, quantity, and function in aged muscle tissue
- Project Gamma
Competitors:
Other companies approach the disease (Duchenne) through gene therapy. They aim to slow down the progression of muscle degeneration. Satellos’s approach (muscle regeneration with small-molecule therapeutics) is unique.
There’s a difference between regeneration and slowing down!
“Satellos is taking an entirely novel approach to the treatment of Duchenne by focusing on stimulating muscle regeneration as opposed to slowing muscle degeneration. While Sarepta and others focus on muscle fiber and dystrophin production, Satellos targets muscle stem cells and reinstates their ability to produce new muscle.”
Potential:
How do markets value an effective drug for this disease??
Sarepta Therapeutics, the largest company developing drugs for Duchenne muscular dystrophy, temporarily lost 5B (USD) of its market value when the drug it developed for Duchenne muscular dystrophy received poor results).
Founders:
Co-founder CEO: Frank Gleeson
Co-founder CDO: Michael Rudnicki
These gentlemen have a history:
Fate therapeutics and Verio Therapeutics
So, it’s not their first rodeo.
The stock is illiquid, with large daily fluctuations. The drug development sector is full of risks!
Insider ownership approx. 15%
Institutional ownership 50%
I would appreciate others’ views on the company.