I came across this tweet related to Novo Nordisk, it might interest at least some readers of the thread. ![]()
Very good Q4 result
Novo Nordisk’s sales increased by 31% in Danish kroner and by 36% at constant exchange rates to DKK 232.3 billion in 2023
" Novo Nordisk to acquire three fill-finish sites from Novo Holdings A/S in connection with the Catalent, Inc. transaction
Bagsværd, Denmark, 5 February 2024 – Novo Nordisk today announced that the company has agreed to acquire three fill-finish sites from Novo Holdings A/S (Novo Holdings) in connection with a transaction where Novo Holdings has agreed to acquire Catalent, Inc. (Catalent), a global contract development and manufacturing organisation headquartered in Somerset, New Jersey (US). Novo Nordisk and Catalent have a long-standing collaboration."
The day will be centred around the Strategic Aspirations. During the day, senior management presentations will cover corporate strategy and purpose & sustainability (ESG), Research and early development, Product Supply as well research and development pipeline and performance within Diabetes care, Obesity care and Rare disease. In addition, Novo Nordisk’s approach to Cardiovascular & emerging therapy areas and financials are presented.
The CMD will include break-out sessions covering Region EMEA, Region China and Data science & Artificial intelligence.
Key highlights of the day are:
- Focus on early-stage pipeline expansion by building on core research capabilities and new technology platforms. Phase 1 data for amycretin, a novel GLP-1 and amylin co-agonist, will also be presented
- Update on scaling of manufacturing capacity and plans to increase patient reach
- Continued growth potential for GLP-1-based semaglutide treatments within Diabetes care and presentation of data from the kidney outcomes trial FLOW
- Update on commercial launches for Wegovy® in the US and International Operations, an update of the obesity research and development pipeline, including additional data from the cardiovascular outcomes trial SELECT and overlap of comorbidities in people with cardiometabolic diseases
- Update on the expanding pipeline within cardiovascular disease
- Expected margin development in the coming years
Myös tällainen merkittävä uutinen: 8.3.24: Novo Nordisk Wegovy® FDA approved in the US Wegovy for cardiovascular risk reduction in people with overweight or obesity and established cardiovascular disease: (FDA) has approved a label expansion for Wegovy® based on a supplemental New Drug Application (sNDA) for the indication of reducing risks of major adverse cardiovascular events (MACE) including cardiovascular death, non-fatal heart attack (myocardial infarction) or non-fatal stroke in adults with either overweight or obesity and established cardiovascular disease (CVD)…The findings from SELECT also showed that over a period of up to five years, risk reductions in MACE were achieved regardless of baseline age, sex, race, ethnicity, body mass index (BMI) and level of renal function impairment. In addition, the label is updated to include data from SELECT showing a risk reduction in cardiovascular death by 15% and a risk reduction of death from any cause by 19%, both compared to placebo1. Further, additional clinical data from SELECT are included in the label.

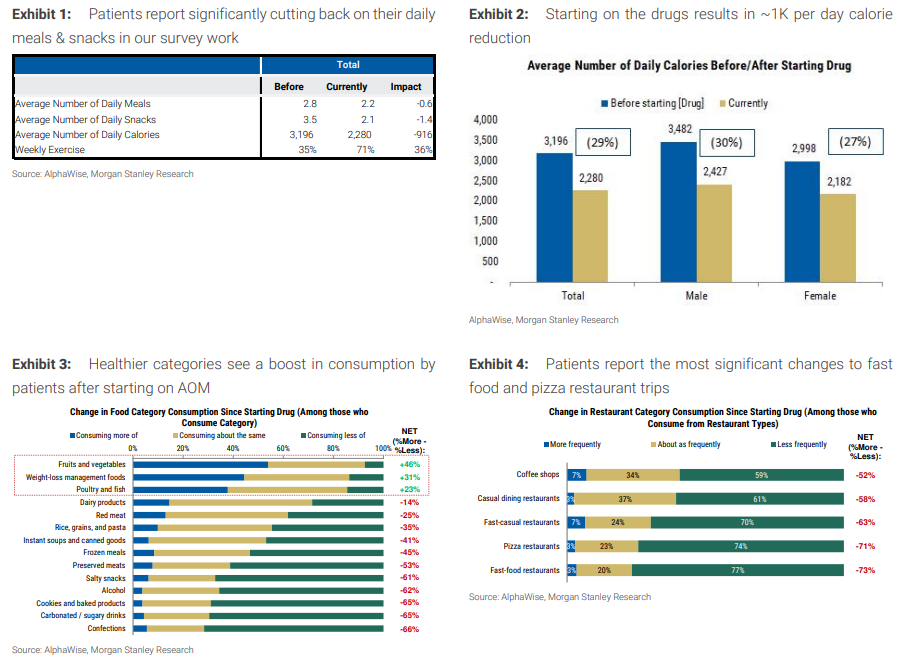
Big changes.
AOM = Anti-obesity medication
@Arimatti_Alhanko since you’ve apparently been following the company more closely, is there a plan to scale up production in a big way so that treating obesity with this could be feasible? The need is likely… huge ![]()
“The doctor suggested the oral gut hormone-mimicking drug Rybelsus.”
Drug sales have indeed had to be restricted as demand has exceeded production capacity.
Novo recently acquired more production capacity by, among other things, buying the American company Catalent.
https://www.cnbc.com/2024/02/05/novo-nordisk-parent-to-buy-catalent-to-expand-wegovy-supply.html
Here is a translation of today’s news from Marketwire/Nordnet:
However, Novo has struggled to keep up with massive demand, and it decided last May to limit access to Wegovy starter doses in the United States to ensure that those who have started treatment can also continue with higher doses.
Since then, however, production has ramped up, and Novo announced in connection with its financial results that it had more than doubled the supply of lower Wegovy doses.
In addition, Novo plans to gradually expand the total supply during the remainder of 2024, Lars Fruergaard Jørgensen said."
.Eikös se mennyt päinvastoin eli Eli Lilly ylitti uusissa resepteissä Novon . Noo kummatkin on tänä vuonna kurssi noussut saman verran.
March 15 (Reuters) - Eli Lilly’s powerful weight-loss drug Zepbound hit 77,590 new prescriptions in the U.S. for the week ending March 8, surpassing Novo Nordisk’s (NOVOb.CO), opens new tab rival obesity medicine Wegovy for the first time since it was launched, according to data from IQVIA.
https://www.reuters.com/business/healthcare-pharmaceuticals/lilly-weight-loss-drug-zepbound-new-us-prescriptions-surpass-wegovy-first-time-2024-03-15/
That’s right, I deleted part of the text, thanks for the correction! For some reason, Google’s direct translation was a bit off in the text I quoted. The source language was Danish.
The text related to demand/production capacity is likely correct, though.
The investment case has changed quite a bit since Novo first appeared in my portfolio ![]() Back then, we were talking about the diabetes market. Sales of the once-weekly injectable semaglutide Ozempic had only just begun, and Rybelsus (the oral version) was the big thing, and it had the potential to become something massive. At that time, Rybelsus wasn’t even priced into the stock yet; it was an extra option. But for some reason, Rybelsus sales have been surprisingly sluggish, especially compared to Ozempic.
Back then, we were talking about the diabetes market. Sales of the once-weekly injectable semaglutide Ozempic had only just begun, and Rybelsus (the oral version) was the big thing, and it had the potential to become something massive. At that time, Rybelsus wasn’t even priced into the stock yet; it was an extra option. But for some reason, Rybelsus sales have been surprisingly sluggish, especially compared to Ozempic.
Today, obesity is the “hot potato.” Before, there was Saxenda, whose sales were really sluggish. The proven benefits were just so small. Then Wegovy (also based on “sema”) was discovered, and its sales are expected to continue with a steep growth curve at least until the even more effective Cagrisema starts to conquer the market. Try keeping up with all this ![]()
Right now, the obesity market seems to be a two-horse race between Novo and Eli Lilly. If I remember correctly, Morgan Stanley’s current estimate is that about 7% of people in the US will be using anti-obesity medication by 2035. That’s about 24 million people on the other side of the Atlantic alone. Additionally, the drug may have an impact on many other markets, such as sleep apnea. Dr. Fatima Cody Stanford, Associate Professor of Medicine and Pediatrics at Harvard Medical School, mentioned somewhere that most CPAP (sleep apnea) patients could get rid of their devices if they managed to lose at least 15% of their weight. And since the drug has had clear effects on users’ appetite and calorie intake, there might be long-term downward pressure on the demand for Resmed’s CPAP devices, for example.
There are two essential reasons why injectable preparations are favored over oral (p.o.) ones:
- When taken orally, the efficacy of semaglutide is lower
- Side effects (GI tract) are more significant
From this, one can draw a quick conclusion that, for example, as a clinician, in an ideal situation, I would always rather try injectable semaglutide for a patient than oral ![]() Of course, preferences vary and some are afraid of needles etc., but a great many are much more satisfied with those injections vs. oral.
Of course, preferences vary and some are afraid of needles etc., but a great many are much more satisfied with those injections vs. oral.
According to conservative(?) estimates, the obesity drug market will be at least USD 100 billion by 2030, which means roughly a 50% CAGR growth from the present. It should be noted that other drugs essential to Novo (diabetes drugs) are also growing at about an 8-12% CAGR over the coming years. Novo will grow at a staggering pace throughout the 2020s, even if it doesn’t increase its market share at all.
And when looking at the pipelines of the current giants regarding obesity treatments, I find it hard to believe that “anything better” in terms of medical treatment would come along even in the 2030s. By this, I mean a drug candidate based on a completely new mechanism of action. What that means for the size of the obesity drug market in, say, 2035 or 2040, considering that people are only getting more obese globally.
In my opinion, Eli Lilly’s pipeline is more promising for the treatment of obesity, but on the other hand, the demand for these is so staggering that I don’t think it will disrupt Novo’s growth for a long time.
On the other hand, a good question is also when will these become commodities? When will the patents expire and all companies put their own products on the market, triggering price competition? Perhaps not for another 10-15 years, but that time will come eventually. It then remains for us investors to weigh how much we are willing to pay for all this excitement right now…
The patents for Rybelsus and Ozempic are valid in the US until 2032 and in Europe until 2031, so there are still a few years left on those. Victoza’s patent has already expired, but it still generates returns. Victoza, however, never reached the same level of hype as Ozempic and Rybelsus. After all, a drug injected once a week or taken orally is much easier to take than a daily injectable.
In the case of Ozempic, demand exceeds supply. The problem itself isn’t that not enough medication is being produced, but rather that the pens (from which the drug is administered) are not available in sufficient quantities.
The potential for these is massive. In my own portfolio, I have both Novo and Eli Lilly. Eli Lilly’s triple-agonist drug is also promising; it has only just entered the market. It doesn’t seem to be available in Finland yet, though.
Novo appears to have a 55% market share in the GLP-1 drug class. Eli Lilly’s pipeline does look more promising than Novo’s, but I do believe Novo will maintain a strong position, even if Eli Lilly’s products take the upper hand in new prescriptions, because an already started and effective medication is rarely switched just “for the sake of it.”
The difference in efficacy between oral and injectable semaglutide is more a matter of dosage. There isn’t a huge difference in side effects either (they are very common in any case).
Regarding investing in Novo Nordisk, the main concern is future competition (the situation in about 5 years?) and the high valuation relative to this. The products are excellent, but competition is coming from Eli Lilly, and there are others eager to enter the market… for example, Pfizer is still developing oral danuglipron, at least for now.
An interesting overview in Fierce Biotech of GLP-1-based and other obesity drugs under development.
This means there are currently a total of 124 candidates in the pipeline, eight of which are already in phase 3. However, this report mentions that not all drugs necessarily go through the entire pipeline; instead, so-called late-breaking candidates can still enter directly into a later phase, much like Wegovy did back in the day.
Some other good reflections on the topic as well. Here is one highlight related to the discussion above.
While Novo, and soon to be Lilly, have a tight grip on the market, they will face tough competition in the years to come. What’s already clear, though, is that GIP/GLP agonists are the future.
Kleinrock likens the obesity rush to what happened with PD-1 inhibitors in oncology. Once an effect was seen, companies raced into the space. But, ultimately, the clinic winnowed out the also-rans until only the strongest meds advanced.
“There’s a lot going on. How many do you see ultimately in a stable market for obesity 10 years from now? It’s not 124,” Kleinrock said.
He suspects GLP-1s aren’t going anywhere for the indication any time soon. He calls them the “alpha dog” of the therapy group. But “not every launch has to pass number one to be on the market,” he added.
The company’s headquarters were a bit on fire, no injuries and the mini-dip was really quick too. No paywall.
The fire does not affect the buildings used for medicine production, a company spokesperson said via email. Novo’s headquarters campus in Bagsvaerd is located just over ten kilometers from central Copenhagen.
Here is Salkunrakentaja’s article on Novo Nordisk. ![]()
Subheadings:
- Wegovy drug sales are growing
- Competitors are aiming for the market

