Hansa Biopharma announces positive full results from 15-HMedIdeS-09 Phase 2 study and comparative analysis of imlifidase in patients with Guillain-Barré Syndrome
Lund, Sweden, 17 December 2024. Hansa Biopharma, “Hansa” (Nasdaq Stockholm: HNSA), today announced positive full results from the 15-HMedIdeS-09 single arm Phase 2 study of imlifidase, a first in class IgG cleaving enzyme, in Guillain-Barré Syndrome (GBS) and an indirect treatment comparison of the 15-HMedIdeS-09 study data to the International Guillain-Barré Syndrome Outcome Study (IGOS), a worldwide prospective study by the Inflammatory Neuropathy Consortium on prognosis and biomarkers of GBS.
Data from the 15-HMedIdeS-09 study demonstrated that severe GBS patients treated with a single dose of imlifidase (0.25 mg/kg) plus intravenous immunoglobulin (IVIg) had rapid overall improvement in functional status including expedited recovery of muscle strength, fast return to independently walking, and a median time to independently walk (e.g., reaching Guillain-Barré Syndrome Disability Scale (GBS DS) 2 or less) by 16 days.
The indirect treatment comparison concluded that patients in the 15-HMedIdeS-09 study treated with imlifidase plus IVIg returned to independently walking 6 weeks sooner when compared to severe GBS patients in the IGOS real-world comparator group treated with IVIg. Additionally, patients in the 15-HMedIdeS-09 study experienced statistically significant improvement across several clinically meaningful measures at multiple time points as compared to the IGOS real-world comparator group including 6.4 times more likely at week 1, and 4.2 times more likely at week 4 to walk independently.
Hitto Kaufmann, Chief R&D Officer, Hansa Biopharma said, “Our Phase 2 study results and the indirect treatment comparison with IGOS are critically important. Together they demonstrate the significant role imlifidase may play in future treatment options for GBS patients. Unlike other molecules, imlifidase can effectively and very rapidly remove IgG through enzymatic cleavage - halting the progression of nerve damage associated with GBS and stopping disease progression. The main goal of improved GBS treatments is to stop nerve damage early, reducing the time of hospitalization and support patients in regaining independence sooner. These findings underscore the role pathogenic IgG plays in severity and progression of GBS, and the clear potential of imlifidase to address unmet need in IgG-driven autoimmune diseases where faster acting treatment options are needed.”
In GBS, IgG is a key driver of inflammatory attacks on peripheral nerves and has been clinically linked to the severity and progression of the disease. Rapid reduction of IgG levels has the potential to benefit GBS patients by depleting pathological IgG antibodies, thereby halting disease progression resulting in faster recovery and less severe disease.1 Improvement in GBS DS is important because it directly affects the clinical outcomes, recovery, and quality of life for patients. Better management of disease severity can help reduce the risk of life-threatening complications, shorten recovery time, prevent long-term disability, lower healthcare costs, and improve overall patient well-being.
Professor Shahram Attarian, Head of Department of Neuromuscular Diseases and ALS, Hopitaux Universitaires de Marseille (APHM), and International Coordinating Principal Investigator in the 15-HMedIdeS-09 Phase 2 study, said, “In the treatment of GBS and subsequent recovery process, early improvement and the ability to walk independently are important clinical milestones as they indicate a return to basic mobility and independence, and to an improved quality of life for patients. This analysis supports the potential role of imlifidase followed by standard of care IVIg as a potentially new treatment option in GBS. These are important results for patients and clinicians in the GBS community.”
Key Results: 15-HMedIdeS-09 Study
The 15-HMedIdeS-09 study included 30 adult patients who were treated with imlifidase plus IVIg. During the study, three patients were re-diagnosed, and the remaining 27 patients received a confirmatory diagnosis of severe GBS and were included in the efficacy analysis.
By the first week, 37% of patients in the 15-HMedIdeS-09 study were able to independently walk and the mean improvement in muscle strength was 10.7 points as assessed by Medical Research Council (MRC) sum score.
The median time to improve by at least one grade in the GBS DS was six days. By eight weeks, 67% of patients were able to walk independently, 40.7% of patients had regained the ability to run, and 37% of the patients had improved by at least three points in the GBS DS. Six months after imlifidase treatment, 63% of patients were able to run or had no functional disability (GBS DS ≤1). Administration of imlifidase was well tolerated in the study.
Key Results: Indirect Treatment Comparison of 15-HMedIdeS-09 Study with Real-World Comparator Group
When compared to the IGOS real-world comparator group (severe GBS patients treated with IVIg, n=754), patients in the 15-HMedIdeS-09 study (severe GBS patients treated with imlifidase in combination with IVIg, n=27) experienced significantly faster improvement in disability as measured by the GBS DS.
Patients in the 15-HMedIdeS-09 study improved by at least one step on the GBS DS, 3 weeks sooner (p=0.002) and returned to independently walking (GBS DS≤2) 6 weeks sooner versus patients in the IGOS real-world comparator group treated with IVIg (p=0.03).
Moreover, patients in the 15-HMedIdeS-09 study were more likely to quickly regain the ability to independently walk than the IGOS real-world comparator group treated with IVIg. At one week, patients in the 15-HMedIdeS-09 study were 6.4 times more likely (odds ratio 95% confidence interval: 2.3-17.5, p<0.001), and at four weeks, 4.2 times more likely (odds ratio 95% confidence interval: 1.6-11.5, p=0.005) to walk independently than those patients in the IGOS real-world comparator group treated with IVIg. Results were matched and weighted for various prognostic factors including time from weakness onset to treatment initiation and baseline value for age, autonomic disfunction, cranial nerve involvement, GBS DS, and MRC sum score.
Hansa is developing novel immunomodulating biologic therapies based on its proprietary, first in class IgG cleaving platform and is focused on IgG driven immune mediated disease where there is high unmet medical need and little to no treatment options. The company has two IgG cleaving compounds. Imlifidase is a first generation, first in class, single dose therapy with proven efficacy and safety. It’s conditionally approved in the EU for desensitization in kidney transplantation. HNSA-5487 is a second-generation molecule with redosing potential with a clinical development path focused on acute exacerbations in neuro-autoimmune disease including myasthenia gravis (MG).
The company plans to publish data from the study and indirect comparison. More information about the study is available at ClinicalTrials.gov under NCT03943589.
Hansa Biopharma will host a telephone conference on 18 December at 14:00 CET / 8:00 AM ET.
Slides used in the presentation will be available online following the call.
https://www.hansabiopharma.com/media/press-releases/2024/hansa-biopharma-announces-positive-full-results-from-15-hmedides-09-phase-2-study-and-comparative-analysis-of-imlifidase-in-patients-with-guillain-barre-syndrome/
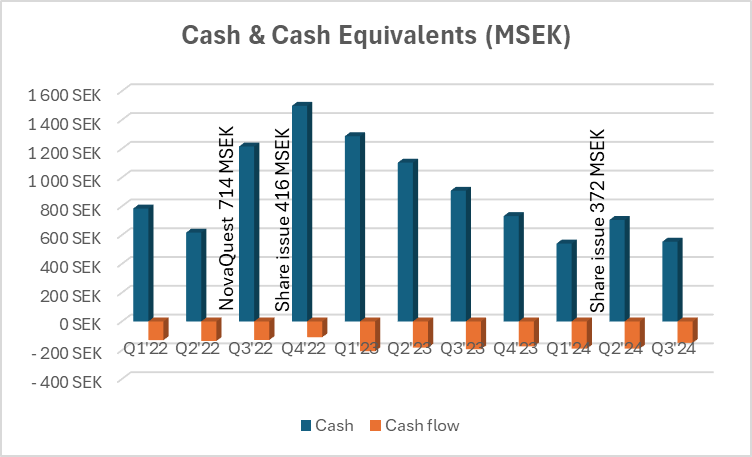
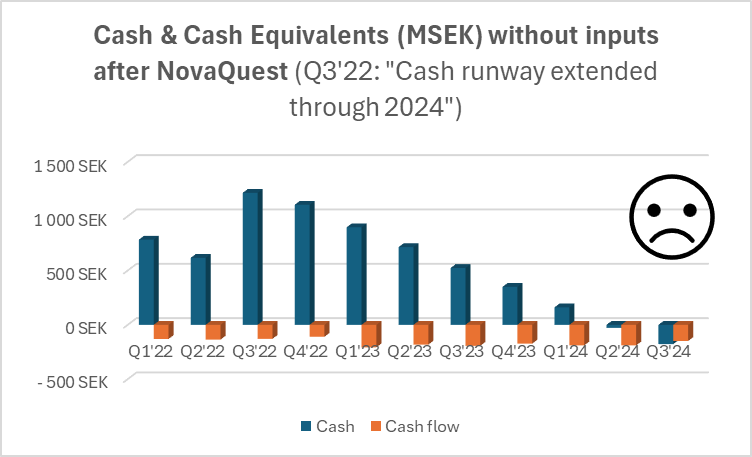
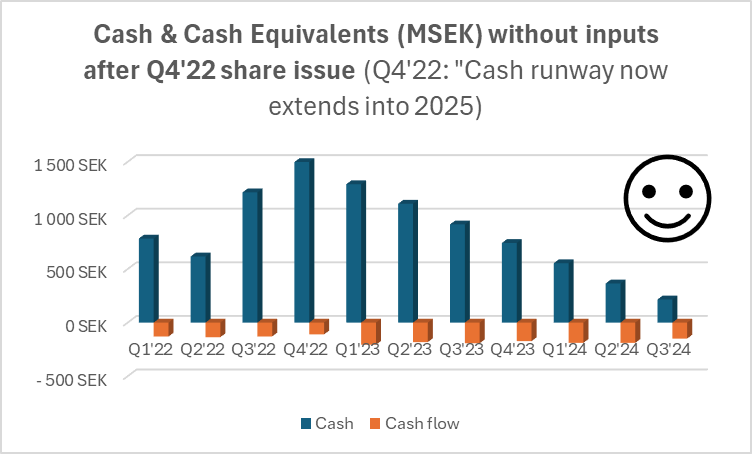
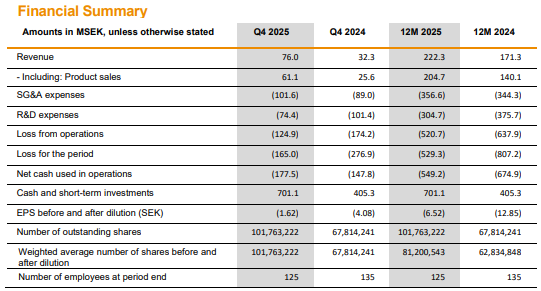
![]() ), Hansa has been quite proactive in its financing arrangements, which is only a good thing. On the other hand, Faron’s emergency issue in the spring was carried out in a retail-investor-friendly way, but Hansa has focused on directed issues. The last one was, of course, at market price, for which the retail investor (tuulipuku) says thanks
), Hansa has been quite proactive in its financing arrangements, which is only a good thing. On the other hand, Faron’s emergency issue in the spring was carried out in a retail-investor-friendly way, but Hansa has focused on directed issues. The last one was, of course, at market price, for which the retail investor (tuulipuku) says thanks ![]() .
.